My wife is at work at a hand surgery pre-op care clinic. It is her birthday. In walks a middle-aged male who is about to have tendon reconstruction after he sustained a crush trauma, and is excited about the possibility of returning to work. He is slightly abrasive and somber, despite the prospect of receiving the surgery.
My wife asks him if he is concerned about the procedure, asks if he is feeling well, asks if there is anything she can do to help. With a quiet manner he states all is well. To make discussion my wife states today is her birthday and she is excited to celebrate when she gets home. He smiles and states today is also his daughter’s birthday. She was born the same year as my wife. He states today is always a rough day for him because she passed away in an emergency room 7 years ago at 21 years of age.
We can never fully understand where those we treat are coming from, their life experiences, or what their home situations, thoughts, fears, dreams, and worries entail; and this was a reminder for me. He stated that his daughter was healthy,woke up without issue on that day, but later developed difficulty breathing and wheezing. She was diagnosed with an acute asthma attack. She had one episode in the distant past but was not on any routine medications. She was given albuterol and experienced a negative reaction to the albuterol and completely stopped breathing. She was brain dead by the time she was intubated. They withdrew care in the emergency department.
After my wife shared this with me, I searched to find any case reports of paradoxical reactions to albuterol. Below are three related cases, however bronchospasm becoming worse with beta-blockers is exceptionally rare.
Case reports of paradoxical bronchospasm to inhaled beta agonists:
- http://www.ncbi.nlm.nih.gov/pubmed/16553105 – Paradoxical bronchospasm: a potentially life threatening adverse effect of albuterol (2006).
- http://www.ncbi.nlm.nih.gov/pubmed/18443029 – Paradoxical response to levalbuterol (2008).
- http://www.ncbi.nlm.nih.gov/pubmed/23173379 – Paradoxical reaction to salbutamol in an asthma patient (2012).
What I think more probable, and possibly related to the above case reports, is acute laryngospasm. The albuterol she received may have further irritated her vocal cords potentially worsening, rather than relieving her vocal cord dysfunction. Both Resus.me and LITFL (Life In The Fast Lane) have very useful articles describing management (see below for links). Here is a brief synopsis:
Laryngospasm
What is it?: a potentially life-threatening closure of the vocal chords (can occur spontaneously). Often misdiagnosed as asthma—especially exercise-induced asthma (more common in white females).
How to diagnose (and differentiate from asthma):
- Stridorous sounds are usually loudest over the anterior neck, beware wheezing sounds transmit throughout the lungs
- Typically, albuterol has minimal to no beneficial effect.
- Subjectively more difficulty on inspiration than expiration
Clues in history: recent exercise, GERD, ENT procedures, or extubation
Common causes & some that are not-so common:
- Post extubation
- Exercise
- GERD
- Medications (e.g., (1) ketamine sedation, incidence 1-2 %; (2) versed (very rarely), which can be reversed with flumazenil)
- Near drowning/ aspiration
- Inhalants (smoke, ammonia, dust, cleaning chemicals)
- Related to anxiety
- Strychnine (plant based poison, sometimes used as a pesticide for birds and rodents, also the poison reportedly used to kill Alexander the Great in 323 BC)
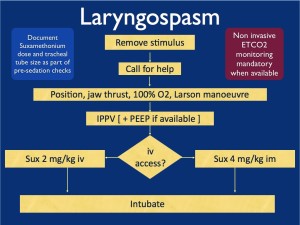
Treatment of laryngospasm:
Initially:
- Jaw thrust with Larson Maneuver
- CPAP/ NIPPV
- Heliox might be helpful if available, (also topical lidocaine can be applied to larynx if available)
If conservative measures fail:
- Low dose propofol (0.1 mg/kg) ~ give 10 mg
- Low dose succinylcholine (AKA: suxamethonium) 0.1-0.5 mg/kg IV
- All else fails: intubation with succinylcholine 1.5 mg/kg IV
- If no IV access, then succinylcholine IM (3-4 mg/kg). Experts advocate IM injection into the tongue.
- Perform chest thrust maneuver immediately preceding intubation to open the vocal cords and allow passage of the ET tube.
- Monitor for negative pressure pulmonary edema—(from patient pulling hard against closed glottis in the setting of acute asphyxia).
Flow chart from Resus.me
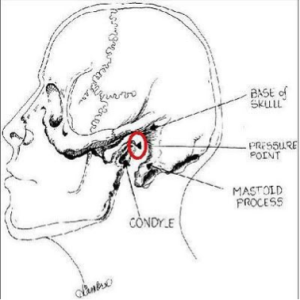
What is the Larson Maneuver? (Published 1998 in Anesthesiology)
It is a manipulation jaw thrust technique targeted at the ‘Larson’s point‘, AKA: laryngospasm notch.
- Place middle finger of each hand in the laryngospasm notch, located behind the lobule of each ear, between ascending ramus of the mandible and the mastoid process.
- Press very firmly inward toward the base of the skull with both fingers
- At the same time lift the mandible at a right angle to the plane of the body (perform jaw thrust).
Reportedly will convert laryngospasm within one or two breath cycles to laryngeal stridor, and in after a couple more breath cycles, to unobstructed respirations. As proposed by Larson, it is likely that the painful stimulus relaxes the vocal cords by way of either the parasympathetic or sympathetic nervous systems through the glossopharyngeal nerve.
Diagram from LITFL
References:
- Resus.Me: http://resus.me/laryngospasm-after-ketamine/
- LITFL (Life In The Fast Lane): http://lifeinthefastlane.com/ccc/laryngospasm/
- UpToDate: https://www.uptodate.com/contents/paradoxical-vocal-fold-motion?source=machineLearning&search=Laryngospasm&selectedTitle=1~150§ionRank=1&anchor=H3#H3
- Larson, Philip, MD. Laryngospasm-The Best Treatment. Anesthesiology. 1998. http://anesthesiology.pubs.asahq.org/article.aspx?articleid=1947036
- Paradoxical bronchospasm: a potentially life threatening adverse effect of albuterol. South Med J. 2006 Mar;99(3):288-9. http://www.ncbi.nlm.nih.gov/pubmed/16553105
- Paradoxical response to levalbuterol. J Am Osteopath Assoc. 2008 Apr;108(4):211-3. http://www.ncbi.nlm.nih.gov/pubmed/18443029
- Paradoxical reaction to salbutamol in an asthma patient. Pneumologia. 2012. Jul-Sep;61(3):171-4. http://www.ncbi.nlm.nih.gov/pubmed/23173379
![Room9er ["Room Niner"]:](https://room9er.com/wp-content/uploads/2020/03/cropped-Screen-Shot-2020-03-08-at-3.16.16-PM.png)