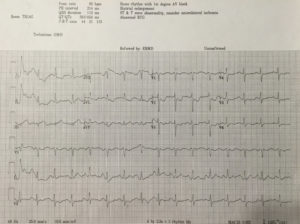
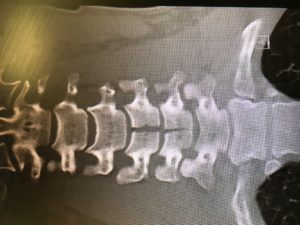
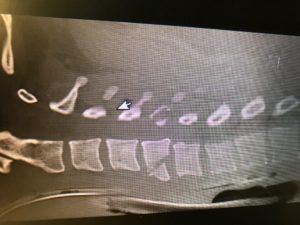
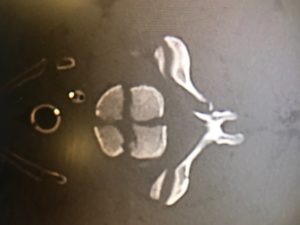
53yo WM rolls in, wee hours of the am. C/o HA, onset around 4 days ago. Wife says “I think he had a seizure in the middle of the night that night” Def hit his head, has a goose egg middle of the forehead. “Had some labs done at my primary yesterday, they called me and told me to come in” No clue as to what labs those might be when asked. Awesome. Medical hx only significant for HTN and GERD. No chronic meds. Admits to drinking around a case of beer a day. Probably explains the not so full history he provided. Nothing else interesting on physical exam and normal vital signs. So I send labs- CBC, Chem13, Mag, CT his head due to the possible seizure.
Annnnd Na 114, K 2.5, CL 75, Bicarb 32, BUN 12, Cr 1.2, Glucose 111, Ca 8.4. CT normal. ETOH and Tox NL. Serum osmolality 259
So what’s going on here? Hyponatremia and seizure- needs fixed right. Normal saline or even hypertonic saline maybe?
Negative ghost rider
Lets look at this a little deeper.
Start with the algorithm

So we will start with the asymptomatic column. Could argue for the severe side due to the seizure, but he is now 4 days out and asymptomatic. Went the euvolemic side of things. No signs of fluid overload clinically and didn’t look dry. Actually nerded out and ordered urine osmolality. 88. So where does that land us?
Beer Potomania syndrome.
WTH is that? Great name. But too much physiology for my brain.
When patients have poor protein and solute (food, electrolytes) intake, such as in chronic alcoholics, they can experience water intoxication with smaller-than-usual volumes of fluid. The kidneys need a certain amount of solute to facilitate free water clearance (the ability to clear excess fluid from the body). A lack of adequate solute results in a buildup of free water in the vascular system, leading to a dilutional hyponatremia.
Free water clearance is dependent on both solute excretion and the ability to dilute urine. Someone consuming an average diet will excrete 600 to 900 mOsm/d of solute. This osmolar load includes urea generated from protein (10 g of protein produces about 50 mOsm of urea), along with dietary sodium and potassium. The maximum capacity for urinary dilution is 50 mOsm/L. In a nutritionally sound person, a lot of fluid—about 20 L—would be required to overwhelm the body’s capacity for urinary dilution.
However, when you don’t eat, the body starts to break down tissue to create energy to survive. This catabolism creates 100 to 150 mOsm/d of urea, allowing you to continue to appropriately excrete a moderate amount of fluid in spite of poor solute intake … as long as you are not drinking excessive amounts of water.
Alcoholics get a moderate amount of their calories via beer consumption and do not experience this endogenous protein breakdown or its resultant low urea/solute level. With low solute intake, dramatically lower fluid intake (about 14 cans of beer) will overwhelm the kidneys’ ability to clear excess free water in the body.
So, we see alcoholics all the time. Why don’t we see this all the time? When you think about it, a pretty low percentage of our EXI all star drinkers are beer drinkers. And most case a day beer drinkers actually have a decent oral food intake. Making this more rare than you originally might think.
So what do we do about it? Classic example of ‘dont just do something stand there!’
Fluid restriction, fluid restriction, fluid restriction.
Back to my case. Admit the patient to my hospitalist, who is half asleep and currently not as excited as I am about hyponatremic management. Orders in for fluid restriction and serial chemistries. A few minutes later, I hear my ER nurse arguing with the floor nurse during calling of report. They are appalled that I am not giving normal saline, and even request hypertonic saline. I politely pick up the phone and discuss the physiology and reason for my treatment plan. We were on the same page by the end of the call.
Fast forward to a couple days later. I come back for another shift, and am checking on my patients from the day before (quick aside, no matter where you are in your career always save the info for 3-4 of your sicker, more interesting patients and look them up on your next shift, by far one of the most high yield learning you can get. And will help you adjust your practice as indicated). I look up the serial sodium results. 114, then 119, 123, then………145. Goooooooo!!!! Assuming I am about to hear from the Kentucky Hammer due to causing central pontine myelinolysis. I talk to the hospitalist- apparently the next shift nurse, after the one I talked to, got her way, and they got an order to blast the guy with normal saline. Hence the huge jump. Patient did have some transient AMS, but was at baseline and neuro intact once levels stabilized. Thank god.
Anyway, interesting case. Beer potomania. cool name, not so cool results. Literature states that central pontine myelinolysis happens in over 20% of these patients due to too rapid correction. So before you pull the trigger on normal saline repletion take a second and scope out the algorithm above. Sometimes the best thing you can do is nothing.
![Room9er ["Room Niner"]:](https://room9er.com/wp-content/uploads/2020/03/cropped-Screen-Shot-2020-03-08-at-3.16.16-PM.png)